Subscribe for Updates
"*" indicates required fields
Navigating the Great Retirement Exodus: Ensuring Manufacturing Continuity with a CMMS and Strong Talent
By: Lisa Bowman, Executive Director of People, Culture and Community In the near future, the American workforce is poised for a significant shift as the…
The Premier Validated Maintenance Management Software Solution for the Life Sciences Industry
Co-authored by Chris Tencza and David Porrill Blue Mountain Regulatory Asset Manager Introduction In the dynamic and highly regulated world of the Life Sciences industry,…
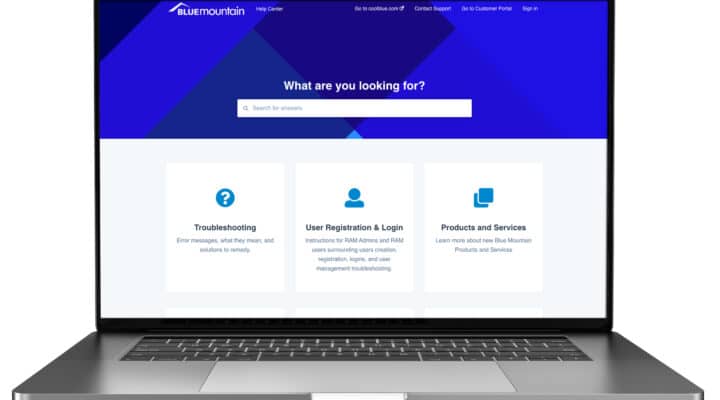
Blue Mountain’s New Help Center: A Central Knowledge Hub
At Blue Mountain, we are committed to our Life Science customers’ success. As part of that commitment, we provide our customers with reliable solutions, and unwavering…
Discover the R4 Training Track at the 2023 Blue Mountain Summit
Ready to enhance your expertise in Blue Mountain RAM? The R4 Training Track at the upcoming 2023 Blue Mountain Summit is the perfect opportunity to…
Explore the R3 Training Track at the 2023 Blue Mountain Summit
Are you ready to take your knowledge of Blue Mountain RAM to the next level? The R3 Training Track at the upcoming 2023 Blue Mountain…
Unlocking Excellence: 7 Reasons to Attend the 2023 Blue Mountain Summit
The Blue Mountain Summit is just around the corner, and if you’re involved in Calibration, Maintenance, Quality, IT, or Operations, this event is an absolute…
Harnessing the Power of AI/ML: Enhancing Regulatory Compliance and Asset Management in the Life Sciences Industry
In the fast-paced world of life sciences, where regulatory compliance and efficient asset management are paramount, the integration of artificial intelligence (AI) and machine learning…
Celebrating the Invaluable Contributions and Transformative Experiences of Blue Mountain’s Summer Interns
At Blue Mountain, we firmly believe in the power of fostering talent and providing enriching experiences for young professionals. This summer, we have had the…
What’s the Difference? FDA 21 CFR Part 11 vs EU Annex 11
This article explains 21 CFR Part 11 and EU Annex 11 regulations for electronic records and signatures in GMP-regulated activities. It covers compliance requirements, benefits,…